 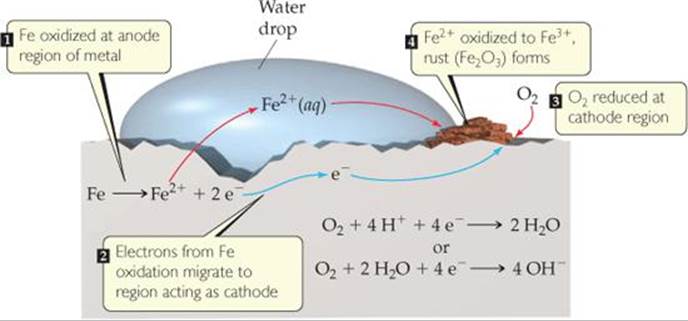 
The investigation of the reaction mechanism via imaging analysis provides valuable information for tuning the calcination chemistry and developing high-energy/power density lithium-ion batteries. In this paper, cathode materials from spent LIBs could be rapidly converted into metal chlorides by NH 4 Cl roasting at 623 K for 20 min. Additionally, it is found that the variations in the reducing power of the transition metals (i.e., Ni, Co, and Mn) determine the local structures at the nanoscale. With the help of a diagram, show the reactions at the cathode and the anode in the manufacture of sodium hydroxide by the Castner-Kellner processPW App Li. Study of reaction characteristics and controlling mechanism of chlorinating conversion of cathode materials from spent lithium-ion batteries J Hazard Mater. Herein, through synchrotron-based X-ray, mass spectrometry microscopy, and structural analyses, it is revealed that the temperature-dependent reaction kinetics, the diffusivity of solid-state lithium sources, and the ambient oxygen control the local chemical compositions of the reaction intermediates within a calcined particle. Reaction conditions: C anode, Pt cathode, constant current 12 mA, 1a (0.5 mmol), 2a (1.0 mmol). The reaction at the cathode is the reduction of the cupric ion to. The electrochemical reactions take place in a lithium-ion battery are summarised as follows: Cathode reaction: LiMO2'Li 1x MO2 xLi+xe (7) Anode. Download scientific diagram Sampling experiment. Although the battery performance depends on the chemical heterogeneity during NRNCM calcination, it has not yet been elucidated. An anode acts as a positive electrode in an electrochemical reaction because electrochemical reactions use electrical energy to transmit a chemical reaction. Electrostatic and Electrochemical Processes 289 + DC Voltage Copper Anode Cathode ++. For the half equation: Al 3+ + 3e- Al, this is described as a. The copper dissolves from the anode and depos. The cathodes and anodes are put into a copper sulfate solution and a current run through the cell. Precise control of the calcination chemistry is therefore crucial for synthesizing state-of-the-art Ni-rich layered oxides (LiNi 1-x-圜o xMn yO 2, NRNCM) as cathode materials for lithium-ion batteries. Reduction reactions occur at the cathode and oxidation reactions occur at the anode during electrolysis. Answer: The electrolytic process essentially takes fairly pure copper-blister copper-and using it as an anode, plates it out onto a pure copper cathode. During solid-state calcination, with increasing temperature, materials undergo complex phase transitions with heterogeneous solid-state reactions and mass transport. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
